A team of U of T engineers is unrolling the mysteries of cancer — literally. They have developed a way to grow cancer cells in the form of a rolled-up sheet that mimics the 3D environment of a tumour, yet can also be taken apart in seconds. The platform, described in a new Nature Materials paper, offers a way to speed up the development of new drugs and therapies and ask new questions about how cancer cells behave.
The drawbacks of studying cancer cells in a traditional petri dish are well known. While cells in a tumour grow in three dimensions, the dish is only two-dimensional. Moreover, cells in the centre of a tumour have less access to oxygen and nutrients than those growing near the surface, close to the blood vessels. These subtle, location-dependent differences have a big impact on cell behaviour, but have proven difficult to replicate in a dish.
In response, tissue engineers have tried to build more realistic 3D models by impregnating porous, sponge-like materials with cells and stacking them like building blocks. Professor Alison McGuigan (ChemE, IBBME) is among them, but she was challenged to think differently about the problem by talking with Professor Radhakrishnan Mahadevan (ChemE, IBBME). (McGuigan and Mahadevan are both members of U of T Engineering’s BioZone.)

“He wanted to do this analysis where you had to collect the cells in less than 10 seconds,” she says. “That was the engineering problem: how do you separate the cells in a very rapid way?” Building blocks wouldn’t do the trick; it would take too long to peel each layer off the structure and would be difficult to keep track of which block came from where.
McGuigan gathered materials from the lab (e.g. filter paper, petri dish, scissors, etc.) and started to think about how to assemble and disassemble them quickly. At one point, she absent-mindedly started wrapping a piece of paper around her finger. “I looked down at it, and realized that’s how you do it,” she says.
Over the next few years, McGuigan and her graduate student Darren Rodenhizer (ChemE 1T3, PhD candidate) built their first prototype. They impregnated a short strip of a porous, paper-like support material with collagen — a gel-like material found in the body — and cancer cells. The whole thing was then bathed in a nutrient-rich culture solution for a day, allowing the cells to adjust to their new environment. Next, the strip was rolled around a metal core, forming an engineered tumour, which was then cultured for a few more days before performing analysis of tumour cell behaviour.
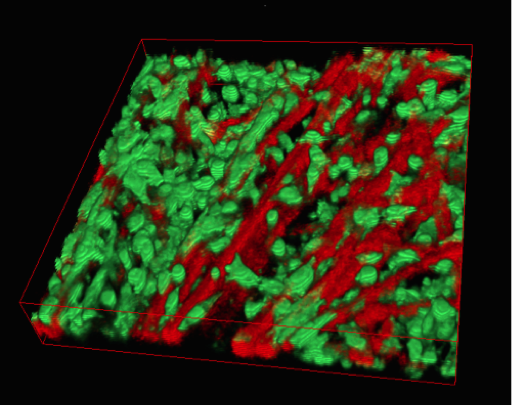
Upon unrolling the device, the team found noticeable differences between the inner and outer layers. “As the oxygen level goes down, the number of dead cells in the layer increases, so the cells are responding to that oxygen gradient,” says Rodenhizer. Those cells that were still alive were shown to behave differently than the surface cells: for example, they more strongly expressed genes associated with low oxygen conditions. Crucially, the changes were gradual and continuous along the length of the strip. “If you had a stack, you could take it apart, but then you’d have all these separate, discontinuous pieces to keep track of,” says Rodenhizer “We have one layer.”
The single-layer design makes it easier for other lab researchers to adopt the process. “It’s simple enough that one could teach an undergrad to do it in a week,” says McGuigan. That makes it a boon to researchers looking to understand what makes cancerous cells in a tumour different from non-cancerous tissue. Exploiting these differences could accelerate the search for drugs that target cancer while leaving healthy cells alone.
Throughout the process, the team has collaborated closely with Christian Frezza at the University of Cambridge and Bradly G. Wouters at Princess Margaret Cancer Centre in Toronto to ensure that the tool enables the types of experimental tests that are most needed by cancer biologists to ask cutting edge questions and translate their findings into benefits to patients.
The technology also holds great promise for the field of personalized medicine. “The idea would be to take a patient’s own cells and create copies of their tumour,” says McGuigan. These copies could then be subjected to various treatments and analysed by the simple unrolling process, providing information about what is likely to work best for that specific patient.
Now that it’s published, McGuigan hopes it will be widely adopted in the research community. “It’s very translatable and transferable to other labs,” says McGuigan. “We definitely want others to use it, because the larger the community, the more applications we will discover.”
Watch Prof. McGuigan describe the technology in her own words at TEDx UofT.
What if we could identify the gene responsible for a baby’s heart defect, grow a piece of her heart on a chip and then test drugs to find the one able to shut down the defective gene?
A decade ago, that scenario seemed as far-fetched as a Mars landing. Now, University of Toronto researchers predict that these stunning achievements are just a few years away, thanks to the Ted Rogers Centre for Heart Research — a powerful collaboration between scientists from U of T’s engineering and medical faculties and physicians from SickKids and the University Health Network. The excitement is building as the Centre celebrates its first anniversary and U of T researchers prepare to move into the MaRS West Tower, which will be the Centre’s new home.
Writer Heidi Singer spoke about the Centre with Professor Craig Simmons (MIE, IBBME), who leads U of T’s collaboration as scientific director for the Translational Biology and Engineering Program.
Learn more about the Translational Biology and Engineering Program.
What is U of T’s role in the Ted Rogers Centre for Heart Research?
Our mission is to help discover the causes of heart failure and engineer therapies. We’re making fundamental discoveries about the mechanisms of cardiac diseases, discovering new biomarkers and building heart tissues with molecules, cells and biomaterials. The overall mission is to reduce hospital admissions due to heart failure by 50 per cent within 10 years. That’s very ambitious, but it can be done. The key is to bring everyone together: medical researchers, doctors, engineers and many other specialties.
Our new space will help make it happen. It’s state-of-the-art, highly integrated and interdisciplinary. We have a whole floor and eventually we’ll have 130 people there. Engineers will be sitting beside biologists who are sitting beside clinicians. This will spark collaborations that would never have happened otherwise.
Can you describe some of the most exciting work at the Centre?
Let’s say a baby is born with heart problems and a doctor at SickKids finds a gene associated with heart failure. Using stem cells from the baby, we can create a model of that child’s heart on a platform that looks like a chip. Then we can do personalized drug testing on the chip before the child ever swallows a pill. The technology exists now. I’m collaborating with a cardiologist who gets the stem cells from the patient and creates heart muscle cells. We put it on the chip. Then we would test drugs on it.
We’re also growing living replacement heart valve tissue in the lab. This could replace our current solution, which is mechanical heart valves that require people to be on blood thinners the rest of their lives. This is important for children because often we replace a valve in babies with fabric. That works for a bit and then they need multiple operations to replace it. What if we can get the baby’s cells from the umbilical cord and grow a heart valve that’s living tissue that will grow with the baby and adapt as the baby grows? We’re doing the same with blood vessels and heart muscle. Instead of using synthetics and other artificial replacements, we could use living replacements that would require one surgery to fully restore function.
We’re already in the early stages, and while we’re several years away from success, all the technology is in place. The demand is there. We know from the surgeons that the current state of the art isn’t great. The challenge now is to create tissues that are comparable to what’s in the body already.
You’re a mechanobiologist. Explain.
This is a field that’s been emerging over the past decade. We’re interested in how mechanical forces in the body affect cell function. We study the heart valve a lot. It opens and closes with every beat of the heart. The valve stretches and bends when blood rushes by. If you have high blood pressure or a malformed valve it causes even more mechanical stress, contributing to valve disease. Most people think of something in the blood, like cholesterol, causing heart disease. But cells react to mechanical forces. One day we’ll make a drug that desensitizes heart cells to mechanical forces.
Dream a bit. What will cardiac care look like in 50 years?
We’ll have injectable biomaterials and molecules to help the heart heal itself. Some animals, like zebrafish, can do it. Whatever gene allows them to do it, maybe we can turn that gene on in humans.
In addition, our ability to grow your exact heart cells in a lab will allow us to conduct drug trials in a Petri dish instead of on an animal. This will give us lightning-fast results. It’s taking some of the guess work out by eliminating something that’s going to fail. Right now drugs are screened in animal models that don’t match up to human biology perfectly. That’s one reason why most promising research falls apart when we get to human trials, which is why it costs so much and takes so long to develop a drug. We could eliminate those false positives sooner and develop personalized drugs for your heart.
Is there anything like the Ted Rogers Centre for Heart Research anywhere in the world?
It’s unique in terms of the number and diversity of people we have. There’s the potential to be in the top five in the world for cardiovascular research and maybe the number one in the world for heart failure. That will come to fruition if we can bring everyone together and synergize. That’s where we’ll really see the potential fulfilled.
Read more about the Ted Rogers Centre for one-year anniversary.
Deep Genomics, the startup company founded by Professor Brendan Frey and his group in The Edward S. Rogers Sr. Department of Electrical & Computer Engineering, has just announced $5 million ($3.7 million USD) in seed financing. The company, launched in July 2015, aims to revolutionize genomic medicine by applying advanced deep-learning computational techniques to unravel mysteries of the human genome.
“Our vision is to change the course of genomic medicine,” said Frey, the company’s president and CEO. “We’re inventing a new generation of deep learning technologies that can tell us what will happen within a cell when DNA is altered by natural mutations, therapies or even by deliberate gene editing.”
The funding round was led by True Ventures of the Bay Area with participation from Bloomberg Beta and other global angel investors, according to TechVibes.
Learn more about Deep Genomics:
The researchers behind Deep Genomics are the first to combine more than a decade of world-leading expertise in machine learning with genome biology. Recently Yann Lecun, director of artificial intelligence at Facebook and a member of the Scientific Advisory Board of Deep Genomics, cited the company’s work in applying artificial intelligence to predict the outcomes of complicated interactions in biological systems as one of the growth areas for AI.
“There is going to be a lot of progress in medicine because of this,” he told New Scientist.
Deep Genomics envisions a future where computers are trusted to predict the outcome of experiments and treatments, long before anyone picks up a test tube. To realize that vision, the company plans to grow its team of data scientists and computational biologists.
Deep Genomics will continue to invent new deep learning technologies and work with diagnosticians and biologists to understand the many complex ways that cells interpret DNA, from transcription and splicing to polyadenylation and translation. Building a thorough understanding of these processes has massive implications for genetic testing, pharmaceutical research and development, personalized medicine and improving human longevity.
Professor Molly Shoichet (ChemE, IBBME) and Natalie Panek (AeroE MASc 0T9) were among the inspiring roster of speakers and performers at this year’s TEDxToronto conference, held October 22.
Shoichet’s talk focused on her research into regenerative medicine and it’s potential to be a game-changer in the treatment of disease. “When I look back on medical treatments [from the past] I’m really happy to be living today,” she said. “But I can’t help but wonder how we will look back on today’s medical treatments. What will we laugh at and ask why?”
Shoichet described her work in three areas — cancer, blindness and stroke — and how her team is going beyond the treatment of the symptoms toward stopping and even reversing these conditions. Her complete talk is now available online:
https://www.youtube.com/watch?v=uRpnmwajwh0&w=560&h=315
Panek discussed “a world we can’t see,” the world of satellites. Though we depend on them for everything from finding our way via GPS to TV signals, the world of satellites are facing a growing problem: only 1 in 7 of those satellites launched since the 1960s is still operational. The increasing amount of “space junk” requires disposal or cleanup, or else it risks damaging working satellites on which we depend.
Panek spoke eloquently about the need for laws to increase the sustainability of our presence in space. “Space is simply a different kind of wilderness that we need to protect,” she said. Her complete talk is also now available online:
https://www.youtube.com/watch?v=GrJG2c0rgqc&w=560&h=315
To view other talks from the conference, including from other members of the U of T community, visit the TEDxToronto website.
This story is Part 1 of an eight-part series, Global Engineering Impact, running throughout fall 2015.
Today, November 19, is World Toilet Day, but if you were able to celebrate it, you should consider yourself lucky. Worldwide, about 2.5 billion people — a third of the global population — have no access to safe sanitation. This lack of hygiene is linked to the spread of many preventable diseases, such as diarrheal diseases that kill more than 500,000 children under the age of five every year.
Since 2011, a team of U of T engineers under the direction of Professor Yu-Ling Cheng (ChemE), director of the Centre for Global Engineering, has been developing a solution. They took up a challenge issued by the Bill & Melinda Gates Foundation to design a toilet that could disinfect human waste without connections to water, sewer or grid power. Its total cost had to work out to less than five U.S. cents per person per day, and it needed to be designed for users in the developing world.
Cheng and her team were among the winners at the first Reinvent the Toilet Fair back in 2012. Since then, with continued support from the Gates foundation, they have been working to develop the disinfection process as well as building and refining their mechanical designs. Writer Tyler Irving caught up with Cheng to learn more about how the project is going.

Your team presented initial proof-of-concept ideas at the original Toilet Fair in 2012, and have been working since then to develop your ideas. Can you update us on your progress?
Things have progressed a lot over the past three years. We created a first integrated prototype, as opposed to the proof-of-concept modules we showed at the time of the first Toilet Fair. That prototype was presented at the second Reinvent the Toilet Fair held in Delhi, India in March 2014.
With continued funding from the Gates foundation, we further refined the design of both the process and the prototype, making it smaller and more energy efficient. We have made a great deal of progress, and have recently been awarded additional support to take us to the point of being able to conduct large scale field trials.
How close do you think you are to a finished product?
I think we’re pretty near completion in terms of designing the technical process. I don’t expect major revisions, but there are things we need to tweak. What we will focus on going forward is to improve robustness and lower costs. In other words, what happens if we run our system for a long time? Does every part still work? Can it stand up to people using – and misusing it? Can we handle variability in user inputs – such as big spikes in the amount of material to process?
How has your design changed since 2012?
At a very high level, the process hasn’t changed very much. We separate solids and liquids, burn the solids to generate heat, and use the heat to dry new incoming solid waste and pasteurize liquid waste. But there have been many advances in the details — these have allowed us to reduce the prototype size by a factor of four — from about 4 m3 to 1 m3, and to lower energy requirement by a factor of five to about 10 watts on average.
How were you able to shrink the size and the energy requirement?
Our first prototype was based on a batch process. We collected a whole day’s worth of waste from a household — which for us was defined as 10 people — and then processed it all at once.
We have since evolved to a continuous process. That means we process small amounts at a steady rate all the time — 24 hours a day, and for as long as we have fuel to feed the process. A system that operates continuously can be much smaller than a batch system, as long as you control the rate at which you’re feeding in new material.
In addition, we only need to provide the energy needed to ignite the process at the beginning of the process — which is a big saving in energy. That initial ignition energy cost is amortized over weeks and months. It becomes a very small energy cost.
We have also improved the way we manage heat. Most significantly, we have eliminated some major inefficiencies in how we dry incoming solids. This sounds like a simple problem, but it has not been easy for us as well as a number of other groups.
Where are you going next?
Going forward, we’re starting to think about how users will interact with it. This is a key challenge: if people require special training to run it, then it won’t meet their needs, so it has to be easy to operate and maintain. It also needs to be robust, and be responsive to changes in input. For a household size of 10 people, we need to process on average about 30 grams of dry fecal mass per hour. But not every household is that big, and not everyone will be home all the time. And we also have to accommodate increased input — parties, weddings, etc. So we have worked out ways to control processing rate over a wide range.
We will do some simulated user studies in our lab in Toronto, probably with the next iteration of the prototype, or something pretty close to it. It’s a squat toilet interface, so not what people here are used to, but it will give us more data on how it responds to variable input.
After that, the next step will be setting up field trials in India or another developing country. That will involve selecting a field site, making sure we have access to users, community approval, access to machine shops for troubleshooting, access to labs for testing, etc. We are planning those studies for August 2016.
What drives you and your team to keep working on this project?
What many of my team members have told me is that they like working on this project because it is so purposeful. The Re-invent the Toilet Challenge set out some very demanding specifications, so it should not be surprising to see some approaches fail. With all the technical advances we have made, I think we have a good chance of succeeding. But even if we don’t get to our final goal, I would much rather not quite reach the final goal working on such a potentially impactful project than doing something else that’s less meaningful.
This interview has been condensed and edited.

Sasha Gollish (CivE MEng 1T0, EngEd PhD Candidate) has been named one of the Top 8 Academic All-Canadians by Canadian Interuniversity Sport (CIS). The award was presented by Governor General David Johnston at Rideau Hall on Monday.
The Academic All-Canadian list recognizes student-athletes who maintain an academic average of 80 per cent or higher while competing for one or more of their university’s varsity teams. Each year, one female and one male from each CIS regional association are selected as Top 8 Academic All-Canadians.
Gollish has already made the list twice, but being selected as one of the Top 8 and meeting with the Governor General gave her a feeling that was “tough to put into words.”
“He talked about three corners of the triangle: athletics, academics and community involvement, and how they re-enforced each other,” said Gollish. “As athletes we are not always recognized for our academic achievements. Being singled out as the top female student-athlete from Ontario was truly an honour.”
Gollish is well-known as one of Canada’s best middle-distance runners. This summer, she captured bronze in the women’s 1,500 metres at the Pan-Am Games in Toronto. She was this year’s CIS athlete of the year, having won five medals at the national championships — including three gold — and helped set a new Canadian record in the 4 x 800m relay.
Academically, Sasha studies with professor Bryan Karney (CivE) in the Engineering Education (EngEd) program. Her research focuses on helping students engage with mathematics. “One of the big jumps that we’ve made in sport in is recognizing the importance of mental engagement and deliberate practice,” says Gollish. She is now trying to bring that coaching experience to the classroom, helping textbook authors and instructors teach in a more concrete way that resonates with the daily experience of engineers.
On top of her academic and athletic commitments, Gollish volunteers with Fast and Female, an organization which supports and empowers girls ages nine to 19 through sport. She is also passionate about making roads safer for recreational road cyclists, serving as a past director of the Ontario Cycling Association.
“On behalf of our department, I warmly congratulate Sasha on the latest of her many accomplishments,” said Professor Brent Sleep, chair of the Department of Civil Engineering. “This honour speaks to the excellence of our student body and the multidisciplinary nature of the programs we offer. Sasha is a great ambassador for the engineering profession.”
Gollish is already focused on her next goal. “I would love to go to the [2016] Olympics in Rio de Janeiro,” she said. “I am training as hard as I can to make that a reality.”
